Disclaimer: Throughout the U.S. and spanning the globe, stem cell research and the science of Regenerative Medicine is gaining acceptance and support as an important part of the future of advanced medicine. The good people who work for State and Federal agencies who regulate clinical medicine are trying the keep pace with the rapid advancement of the field. Thus, Federal and State laws and regulations are mercurial, and constantly evolving. Treatments that are allowed today, might be illegal next week. Unfortunately therefore, you might have to leave the U.S. to find the preferred treatment you are seeking. For questions, feel free to call us at: 949-428-4500 or use our online consultation form.
Every cell in our body comes from the original stem cells that comprised the embryo we once were.
Everything starts with a sperm and egg, which fertilize each other by merging into a single cell thereby becoming the first stem cell from which our entire body will be created. At that point the merged cell starts dividing into 2 cells, then 4, 8, 16, 32 cells etc.
It all starts with a sperm and an egg becoming the first stem cell from which our entire body will be created.
Stem Cell Differentiation Process
Stem cells are a type of cell that has the ability to self-renew and differentiate and can be differentiated into specialized cellular populations. Stem cell differentiation allows stem cells to transition from a unspecialized state to a functionally specialized state, resulting in the formation of various tissues and organs in the body.
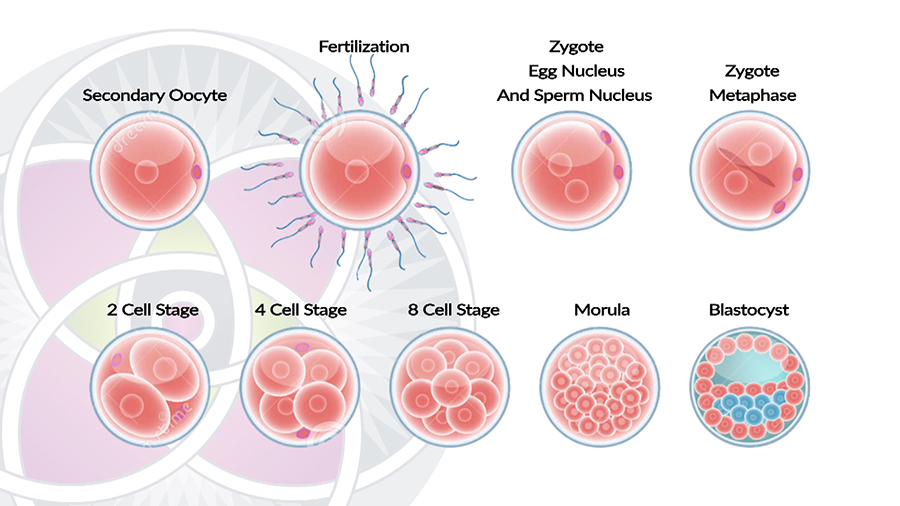
Early stages of embryo development – Pre-implantation in uterus
Totipotent Stem Cells
In these early stages, every one of these cells can become any cell in the human body. These are called “Totipotent” stem cells (TSCs). Because totipotent stem cells can evolve into complete animals, they are used in biological fields such as regenerative medicine and conservation.
Maintaining the developmental totipotency and self-renewal potential of cells is challenging, creating a key hurdle to TSC research. Fortunately, a breakthrough in the study of induced pluripotent stem cells (iPSCs) returning to their totipotent state has resulted in the development of multiple TSCs and the start of a new wave of stem cell research.
As the original stem cells continue to divide, they start changing, a process called “Cell Differentiation” by following three different developmental pathways or “lineages”.
The study of embryonic stem cell cell differentiation decisions has far-reaching implications for regenerative medicine and other bio-medical sciences. Several research groups have extensively examined the regulatory mechanisms controlling stem cell differentiation.
Cell differentiation involves multiple biological components, signaling pathways, and epigenetic alterations. The differentiation of embryonic stem cells has been studied using mathematical models but the basic mechanisms of cell differentiation and lineage reprogramming remain unknown.
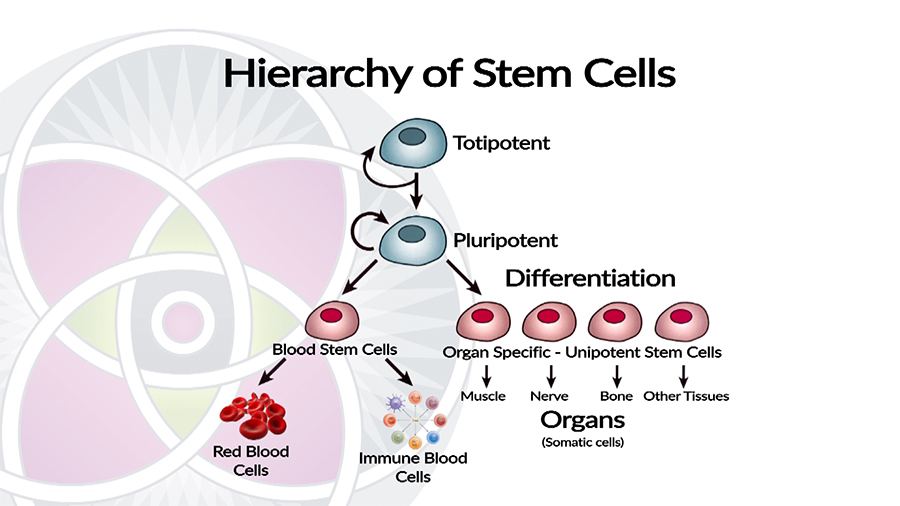
Hierarchy of stem cell differentiation into different organs
Pluripotent Stem Cells
In fact, in the adult human body there are approximately 200 different types of cells all of which fall under these three major categories: Cells that emanate from the endoderm lineage, mesoderm lineage and the ectoderm lineage. Each of these lineages can be identified in the very young embryo, within just a few days of conception.
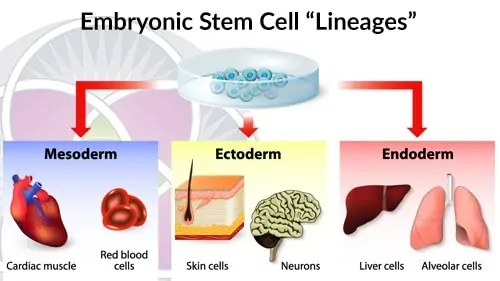
The three major “lineages” of embryonic stem cells.
At this stage all the cells of the embryo are still stem cells, but these cells are limited to developing into the tissues associated with their specific lineage. Thus they are no longer defined as “totipotent” but rather as stem cells with a limited potential of differentiation and are called “pluripotent” stem cells (PSCs).
PSCs are distinguished by the qualities of self-renewal and potency, the former referring to the cell’s ability to multiply and the latter to differentiate into specialized cell types originating from one of the three basic germ layers.
Eventually as these “pluripotent” stem cells undergo further differentiation, they will lose even more potential of differentiation and become “multipotent” (MSCs.
Multipotent Stromal Cells
Multipotent stromal cells (MSCs) are essential for tissue growth, maintenance, function, and regeneration. They can differentiate along various connective lineages, but unlike most other stem/progenitor cells, they can also perform a range of other jobs while still developing. MSCs operate as damage sensors, boosting regeneration in response to injury by releasing trophic factors and extracellular matrix (ECM) molecules, and contributing to fibrotic reparative processes when regeneration fails. Tissue-specific MSC identity, fate(s), and function(s) are being determined providing unprecedented insight into the secret lives of tissue-resident MSCs.
As the embryo continues to develop, its cells choose a path of development down one of those three lineages that lead to specific organs with specific functions. This is called cell “differentiation” that leads to the creation of specific tissues with specific functions, such a nerve tissue, or skin, bone or blood tissue etc. As these early stem cells choose a lineage, they lose their ability to become any tissue, and retain the ability to become just those tissues associated with that specific lineage.
Eventually, every organ and tissue in the adult human will contain a certain percentage of stem cells that are responsible for simply replacing the cells of only that organ that die and need to be replaced.
Unique Characteristics of Stem Cells
Stem cells differ from the other adult kinds of cells that make up all the organs and tissues of the body. All stem cells, regardless of their lineage, have two general properties:
- They are capable of dividing and renewing themselves for long periods, called “self-renewal”
- They are unspecialized in that they do not YET have a specific function… However, when called upon they divide and differentiate into a specific organ cell that does have specific physiological functions.
Self-Renewal Properties of Stem Cells
Stem cells are capable of dividing and renewing themselves for long periods. In contrast adult organ cells – also known as Somatic Cells, such as muscle cells, blood cells, or nerve cells— have a limited number of times they can replicate themselves.
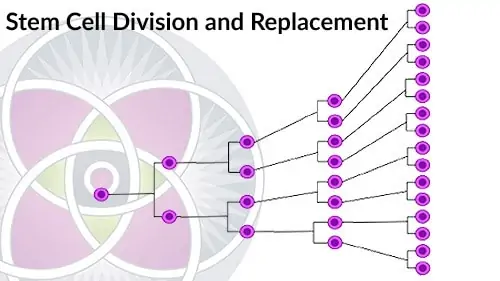
Stem cells are capable of dividing and renewing themselves for long periods.
Eventually every adult somatic cell will age and lose its ability to function at peak efficiency. When this happens, it will undergo “programmed cell death” or “apoptosis”. At this point the dying cell will be replaced from a new cell generated from a local stem cell.
Proliferation (Long-term Self Renewal) of Stem Cells
Conversely, stem cells may replicate themselves essentially throughout a human’s entire lifetime, (proliferate).
In a laboratory under the correct nourishing conditions, a handful of stem cells can proliferate and generate millions of identical stem cells in just a few short weeks, and can essentially continue proliferating for years. This is called long-term self-renewal.
It is also evident that embryonic stem cells have a much more robust ability to self-renew, in comparison to adult stem-cells. The fact is, as we age, the amount of stem cells we have dramatically diminishes. Therefore, our adult stem cells have a limited capacity of long-term self-renewal, and they do eventually die.
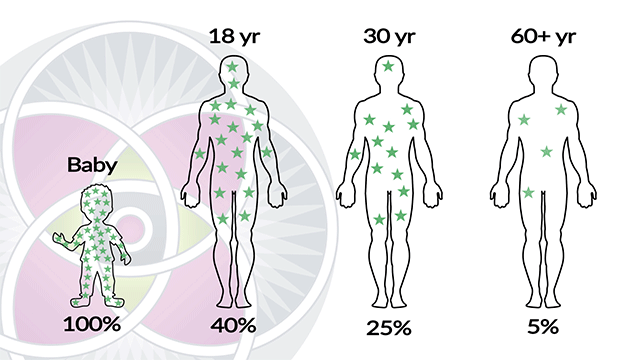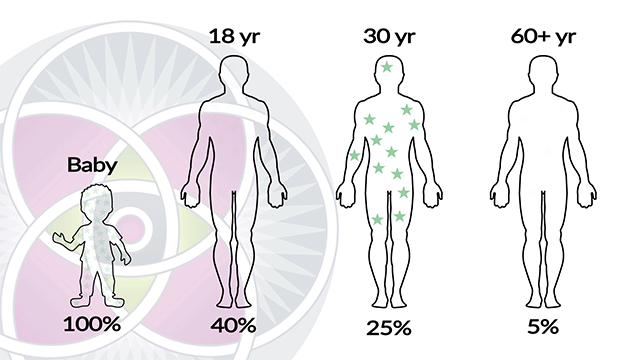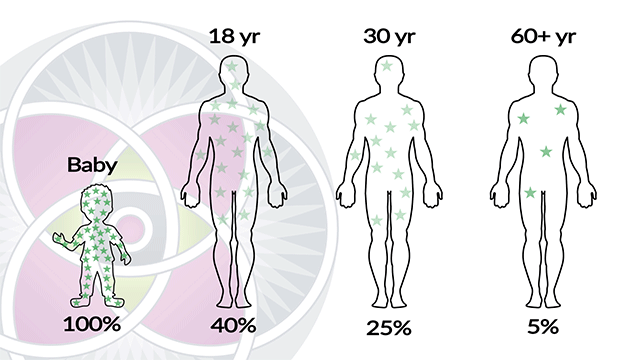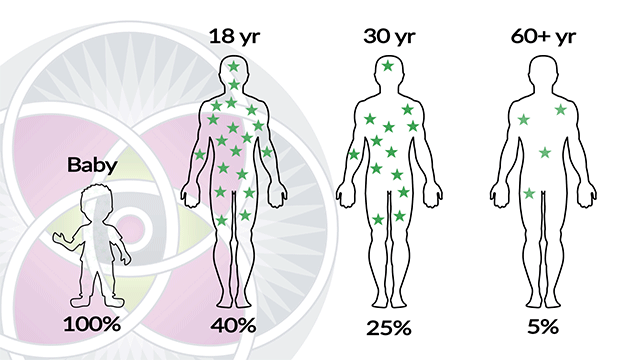
Percentage of stem cells decrease and lose vitality as we age.
Scientists are trying to understand two fundamental properties of stem cells that relate to their long-term self-renewal:
- Why can embryonic stem cells proliferate in laboratory conditions for a year or more without differentiating, but most adult stem cells cannot; and
- What are the factors in living organisms that normally regulate stem cell proliferation and self-renewal?
The hope is that discovering the answers to these questions may reveal two important things:
- Understand how to keep our adult stem cell population large and active.
- What goes wrong with cell proliferation when abnormal cell division occurs that leads to cancer?
Stem Cells are Unspecialized
One of the fundamental properties of a stem cell is that it does not have tissue-specific structures that allow it to perform specialized physiological functions. For example, a stem cell cannot work with its neighbors to cleanse blood (like a liver cell), and it cannot transport oxygen (O2) like a red blood cell.
However, under the right condition, specific physiological signals will stimulate unspecialized stem cells to “differentiate” into specialized cells, including liver, heart muscle cells, blood cells, or any adult organ cells (somatic cells).
The specific factors and conditions that stimulate stem cells to remain unspecialized as well as those factors that stimulate them to undergo Cell-differentiation are of great interest to physicians and scientists. After all, given the fact that human embryonic stem cells give rise to every cell in the adult human, maybe we can learn how to grow entire new and healthy organs in a laboratory as a replacement to a diseased organ.
Stem Cell Cellular Differentiation
When called upon by specific physiological signals and conditions, Stem cells which are unspecialized can give rise to specialized cells (Fig. 4). This process is called cellular differentiation. Cellular differentiation usually requires several stages, during which the cell becomes more specialized at each step.
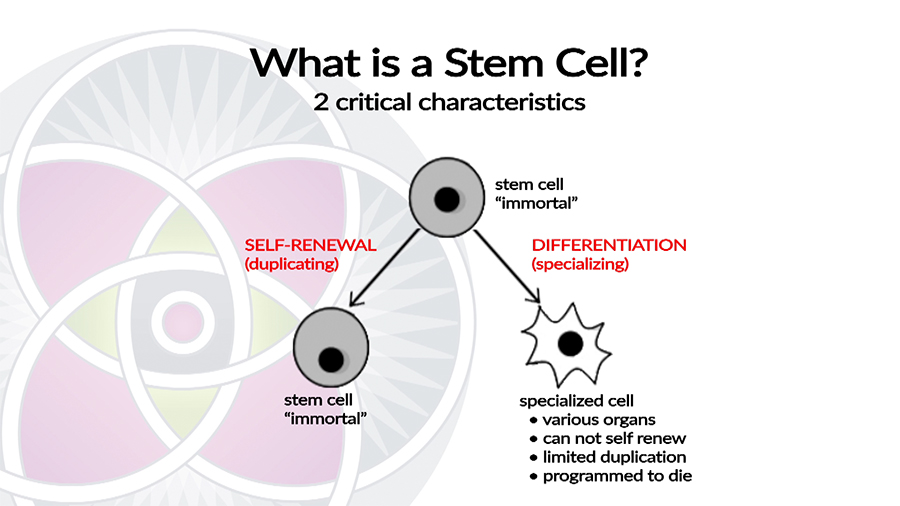
Stem Cells self replicate and generate new specialized organ (somatic) cells.
The Process of Cellular Differentiation
Both internal signals and external conditions contribute to the conditions that give rise to cellular differentiation. Of course many of the internal signals are controlled by the genes of each cells, which are encoded in the cell’s DNA. This is called Gene-Expression.
The external signals that influence cell differentiation include chemicals secreted by other cells such as growth factors, cytokines and lymphokines, as well as physical contact with neighboring cells. All of these influences are inherent properties of the microenvironment in which cells live.
Epigenetics Studies Influences on Stem Cell Differentiation
To the extent that these internal and external influences effect Gene-Expression during stem cell differentiation, has given rise to a new science of Epigenetics. Epigenetics studies how environmental factors, both internal and external control which genes are expressed.
Basically, we now know that cells can acquire epigenetic marks that control DNA expression in the cell and that can be passed on through multiple generations of cells.
Questions About Stem Cell Differentiation Remain
There are still many questions about stem cell differentiation that are unanswered.
For example, are the external and internal signals for cell differentiation similar for all kinds of stem cells?
For instance, can we take a lipid stem cell and a bone stem cell (hematopoietic stem cell) and make them both differentiate into a nerve cell?
Can any adult stem cell become any kind of organ cells?
What are the specific signals that promote differentiation into specific tissue types?
The answers to these questions might lead stem cell researchers to develop novel ways to control stem cell differentiation under laboratory conditions, whereby we can grow cells or tissues that are used for stem cell therapies in treating diseased or damaged organs.
Learn More About the Science of Stem Cells
References - Read the Science Behind Stem Cells...
Everything on our website comes from from reputable publications, books and scientific journals, most of which are available on PubMed and other government websites. These include Meta-Analysis’, Randomized Controlled Trials, Clinical Trials, Systematic Reviews, Books and Documents. We encourage you to read the science, in order to separate fact from fiction, so that you can arrive at a full understanding of what is best for your body. We would be honored to be a part of that educational journey with you.
- Herranz AS, Gonzalo-Gobernado R, Reimers D, Asensio MJ, Rodríguez-Serrano M, Bazán E. Applications of Human Umbilical Cord Blood Cells in Central Nervous System Regeneration. 2010 Mar;5(1):17-22.
- Liu L, Michowski W, Kolodziejczyk A, Sicinski P. The cell cycle in stem cell proliferation, pluripotency and differentiation. Nat Cell Biol. 2019 Sep;21(9):1060-1067. doi: 10.1038/s41556-019-0384-4. Epub 2019 Sep 2. PMID: 31481793; PMCID: PMC7065707.
- Zarrabi M, Mousavi SH, Abroun S, Sadeghi B. Potential uses for Cord Blood Mesenchymal Stem Cells. 2014 Winter;15(4):274-81.
- Kabataş S, Civelek E, İnci Ç, Yalçınkaya EY, Günel G, Kır G, Albayrak E, Öztürk E, Adaş G, Karaöz E. Wharton’s Jelly-Derived Mesenchymal Stem Cell Transplantation in a Patient with Hypoxic-Ischemic Encephalopathy: A Pilot Study. 2018 Oct;27(10):1425-1433.
- Sadlik B, Jaroslawski G, Puszkarz M, Blasiak A, Oldak T, Gladysz D, Whyte G. Cartilage Repair in the Knee Using Umbilical Cord Wharton’s Jelly–Derived Mesenchymal Stem Cells Embedded Onto Collagen Scaffolding and Implanted Under Dry Arthroscopy. 2018 Jan;7(1):e57–e63.
- Kim DW, Staples M, Shinozuka K, Pantcheva P, Kang SD, Borlongan CV. Wharton’s Jelly-Derived Mesenchymal Stem Cells: Phenotypic Characterization and Optimizing Their Therapeutic Potential for Clinical Applications. 2013 May 31;14(6):11692-712.
- Hutchings G, Janowicz K, Moncrieff L, Dompe C, Strauss E, Kocherova I, Nawrocki MJ, Kruszyna Ł, Wąsiatycz G, Antosik P, Shibli JA, Mozdziak P, Perek B, Krasiński Z, Kempisty B, Nowicki M. The Proliferation and Differentiation of Adipose-Derived Stem Cells in Neovascularization and Angiogenesis. Int J Mol Sci. 2020 May 27;21(11):3790. doi: 10.3390/ijms21113790. PMID: 32471255; PMCID: PMC7312564.
- Nagamura-Inoue T, He H. Umbilical Cord-Derived Mesenchymal Stem Cells: Their Advantages and Potential Clinical Utility. 2014 Apr 26;6(2):195–202.
- Kalaszczynska I and Ferdyn K. Wharton’s Jelly Derived Mesenchymal Stem Cells: Future of Regenerative Medicine? Recent Findings and Clinical Significance. 2014 Dec.
- Vasanthan J, Gurusamy N, Rajasingh S, Sigamani V, Kirankumar S, Thomas EL, Rajasingh J. Role of Human Mesenchymal Stem Cells in Regenerative Therapy. Cells. 2020 Dec 31;10(1):54. doi: 10.3390/cells10010054. PMID: 33396426; PMCID: PMC7823630.
- Costanzo, M et al. Low ozone concentrations stimulate cytoskeletal organization, mitochondrial activity and nuclear transcription. European journal of histochemistry : EJH vol. 59,2 2515. 21 Apr. 2015.
- Serra MEG, Baeza-Noci J, Mendes Abdala CV, Luvisotto MM, Bertol CD, Anzolin AP. The role of ozone treatment as integrative medicine. An evidence and gap map. Front Public Health. 2023 Jan 16;10:1112296. doi: 10.3389/fpubh.2022.1112296. PMID: 36726625; PMCID: PMC9885089.
- Clavo B, Borrelli E. Editorial: Ozone in medicine: Biochemical background, physiological modulation and clinical applications. Front Physiol. 2023 Feb 6;14:1112860. doi: 10.3389/fphys.2023.1112860. PMID: 36814483; PMCID: PMC9939878.
- Sucuoğlu H, Soydaş N. Efficacy of ozone therapy as an add-on treatment in fibromyalgia: A randomized double-blind placebo-controlled study. J Back Musculoskelet Rehabil. 2023;36(2):357-366. doi: 10.3233/BMR-210368. PMID: 36530072.